
McGill Univ. Libraries and the generosity of Dr. Richard L. Tomlinson
 |
Brought to you by: McGill Univ. Libraries and the generosity of Dr. Richard L. Tomlinson |
|||
|
|
|
| Bioorganic
& Medicinal Chemistry Letters Volume 10, Issue 20, 16 October 2000, Pages 2325-2327 |
| ||||
|
|
| ||||
Design and synthesis of a novel water soluble benzotetrazepinone
Beatrice Dumont-Hornebeck, Yi Ning Strube, Daniela Vasilescu and Bertrand
Jacques Jean-Claude![]() ,
, ![]()
Abstract
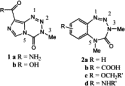
In order to confer water solubility to the benzotetrazepinone ring system,
the synthesis of 12 was undertaken. The design and synthesis of
12 were based upon previously established structural
requirements for the stability of the 1,2,3,5-tetrazepin-4-one ring system.
Tetrazepinone 12 was extremely water soluble and was 10-fold
more potent than its imidazo-1,2,3,5-tetrazin-4-one counterpart
1a, against the human MCF-7 breast cancer cell line.
Tetrazepinones of type 2a
are a novel class of antitumour agents which are more potent than temozolomide
1a
(a strong alkylating agent) against alkylating-agent-resistant Mer+ tumour
cells. [1
and 2]
In contrast to 1a,
Maxam–Gilbert assays show that tetrazepinones are weak alkylating agents that
create barely detectable levels of guanine-N7 alkylation in DNA. [3]
Alkaline sucrose density–gradient sedimentation studies indicate that despite
their weak alkylating activity, tetrazepinones induce significant levels of
single strand breaks in human tumour cells. All the results obtained thus far
suggest that tetrazepinones may damage DNA by a novel mechanism.
 |
Display Full Size version of this image (3K) |
1.
Studies designed to mimic the interactions of tetrazepinones with nucleosides under physiological conditions have been extremely limited by the poor water solubility of the existing compounds. During the development of the chemistry of this novel class of heterocycles,[4, 5, 6, 7, 8 and 9] the marked instability of the 1,2,3,5-tetrazepin-4-one moiety precluded the synthesis of compounds containing functional group at the 3- and 5-positions: bulky substituents at these positions destabilize the 1,2,3,5-tetrazepin-4-one ring, whereas electron-withdrawing groups at the aromatic ring stabilize it. [5] In addition, tetrazepinones are rapidly converted to their corresponding 2-diazoniumphenyl urea precursors at acidic pH. [7 and 9] Therefore, benzoic acid-fused tetrazepinones like 2b, which could be used for coupling with a water soluble amine, cannot be synthesized. In contrast, imidazotetrazinone 1b is stable enough to be converted to its corresponding acyl chloride for further coupling with amine-containing biomolecules. [10 and 11] To circumvent the problems associated with the instability of the tetrazepinone ring system, we designed compound 12, which contains all the structural requirements for a stable molecule. The 3- and 5-positions are substituted with methyl groups and the water soluble 4-aminoethylmorpholine moiety is connected to the aromatic moiety via an electron-withdrawing carboxamido (in order to preserve the electron-withdrawing character of the benzene ring). Ring closure to the 1,2,3,5-tetrazepinone ring was planned to be performed at the last step of the total synthesis.
The preparation of 12 proceeded according to Scheme 1. Acid 3 was treated with aqueous methylamine in a pressure vessel kept at 160 °C to give 4 which was converted to the corresponding acyl chloride 5 by treatment with thionyl chloride. Esterification with trimethylsilyl ethanol and hydrogenation gave 6, which was treated with benzylchloroformate to provide a mixture of 7 and 8 in a 2:1 ratio. Isomers 7 and 8 were extremely difficult to separate by columnn chromatography. Therefore, the mixture was treated with methyl isocyanate to provide the corresponding ureas. Separation of the mixture on silica gel provided the desired urea 9. After removal of the trimethylsilylethyl group by treatment with tetrabutyl ammonium fluoride (1.5 equiv) in tetrahydrofuran, the resulting acid was refluxed in thionyl chloride for 30 min to give acyl chloride 10 in quantitative yield. Coupling of 10 with 4-(2-aminoethyl)morpholine provided the corresponding amide 11a which was converted to amine 11b by catalytic hydrogenation. The latter amine was diazotized with 15N NaNO2 and cyclized at pH 9 to give the desired tetrazepinone 12.
Display Full Size version of this image (12K) Scheme 1. (a) aq CH3NH2 (40%), sealed (160 °C), 70%; (b) SOCl2 (neat) reflux, 100%; (c) 2-(trimethylsilyl)ethanol (1.1 equiv), pyridine (1 equiv), 72%; (d) H2/Pd–C, methanol, 90%; (e) K2CO3, PhCH2OCOCl; (f) CH3NCO in CH2Cl2, 35%; (g) tetrabutylammonium fluoride (1 M in THF), 82%; (h) SOCl2 (neat), reflux, 100%; (i) K2CO3, 4-(aminoethyl)morpholine, 98%; (j) H2/Pd–C, methanol, 90%; (k) NaNO2/H+ and K2CO3, 51%; Z=carbobenzyloxy.
The structure of 12 was confirmed by 1H and 15N NMR spectroscopy, chemical ionization (CI) mass spectrometry and elemental analysis.[12] Tetrazepinone 12 was extremely soluble in water. In 1H NMR, the 3-methyl group appeared as a doublet at 3.4 (3J 15NH=2.8 Hz) in CDCl3 and as a singlet at 3.1 ppm in D2O. In 15N NMR, N2 appeared at 69.16 ppm in H2O (Fig. 1a). The peak assignment was based on literature values.[4 and 9] These data provide the first evidence for a ring-closed tetrazepinone structure in aqueous medium.
Display Full Size version of this image (6K) Figure 1. (a) 15N NMR spectrum of 12 in D2O; (b) dose–response curve of MCF-7 cells exposed to temozolomide and 12 for 72 h.
The CI mass spectrum showed a minor MH+−29 (15NN) peak and a large MH+−MeNCO peak. The molecular ion could not be detected by mass spectrometry using chemical ionization, however the characteristic MH+−N2 and MH+−MeNCO peak were observed.[4 and 5]
Tetrazepinone 12 was tested against the resistant MCF-7 breast tumour cell line using the sulforhodamine B assay. [13] As shown in Figure 1b, 12 (referred to as MOPH) was 10-fold more potent than temozolomide against the Mer+ breast tumour cell line MCF-7 [IC50 (temozolomide): 800 μM, IC50 (12): 80 μM].
The limitations imposed by the instability of the 1,2,3,5-tetrazepinone ring
to the synthesis of conjugated structures can be circumvented by carefully
designing a conjugated 2-aminoaromatic urea, which can be cyclized by
diazotation at the last step of the total synthesis. The availability of this
first water soluble tetrazepinone will stimulate further studies on the
interactions of tetrazepinones with water soluble biomolecules and thereby
significantly improve our understanding of the mechanism of action of this novel
class of cytotoxic agents.
Acknowledgements
We thank the National Cancer Institute of Canada (NCIC) and the Fonds pour la formation des Chercheurs et l'Aide à la Recherche (FCAR) for financial support.
References
1. B.J. Jean-Claude, A. Mustafa, A.J. Watson, Z. Damian, D. Vasilescu, T.H. Chan and B. Leyland-Jones. J. Pharmacol. Exp. Ther. 288 (1999), p. 467.
2. B.J. Jean-Claude, A. Mustafa, J. De Marte, R. Yen, T.H. Chan and B. Leyland-Jones. Can. J. Infect. Dis. 6 (1997), p. 465C.
3. B.J. Jean-Claude, A. Mustafa, Z. Damian, J. De Marte, R. Yen, T.H. Chan and B. Leyland-Jones. Br. J. Cancer 76 (1997), p. 467. View Record in Scopus | Cited By in Scopus (5)
4. B.J. Jean-Claude and G. Just. J. Chem. Soc., Perkin Trans. 1 (1991), p. 2525. Full Text via CrossRef
5. B.J. Jean-Claude and G. Just. Heterocycles 48 (1998), p. 1347. View Record in Scopus | Cited By in Scopus (7)
6. C.I. Williams, M.A. Whitehead and B.J. Jean-Claude. J. Mol. Sruct. (Theochem.) 392 (1997), p. 27. Abstract | Abstract + References | PDF (1186 K) | View Record in Scopus | Cited By in Scopus (3)
7. C.I. Williams, M.A. Whitehead and B.J. Jean-Claude. J. Org. Chem. 62 (1997), p. 7006. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (8)
8. B.J. Jean-Claude, J.F. Britten and G. Just. Acta Crystallogr. C49 (1993), p. 1070. Full Text via CrossRef
9. B.J. Jean-Claude and C.I. Williams. Magn. Res. Chem. 36 (1998), p. 87. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (5)
10. S.A. Clark, B. Deans, M.F.G. Stevens, M.J. Tisdale, R.T. Wheelhouse, B.J. Denny and J.A. Hartley. J. Med. Chem. 38 (1995), p. 1493.
11. K.R. Horspool, M.F.G. Stevens, C.G. Newton, E. Lunt, R.J.A. Walsh, B.L. Pedgrift, G.U. Baig and F. Lavelle. J. Med. Chem. 33 (1990), p. 1393. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (19)
12. Experimental data for 12: Urea 11b (95 mg, 0.2 mmol) was dissolved in 2.4 N HCl (10 mL) and a solution of 27% NaNO2 (1 mL) [50% enriched with 15N NaNO2] was added dropwise at 0 °C. After 30 min, the solution was extracted three times with 25 mL portions of CH2Cl2. The aqueous layer was separated and neutralized with saturated sodium bicarbonate, after which it was further re-extracted three times with 25 mL portions of CH2Cl2. The organic layer was removed and dried over anhydrous potassium carbonate and evaporated to give 12 (50 mg, 51%) as a pale brown powder: mp 125–130 °C (effervescence); δH (CDCl3) 7.95 (1H, d, J=2 Hz, Ar), 7.93 (1H, dd, J=8.6 Hz, J=2, Ar), 7.26 (d, 1H, J=8.6, Ar), 7.05 (1H, br s, NHCO), 3.78 (4H, m, (CH2)2), 3.59 (2H, br q, NCH2CH2NHCO), 3.42 (3H, d, 3J 15NH=2.8 Hz, 15NNCH3), 3.31 (3H, s, N(CO)CH3), 2.64 (2H, t, J=4.5 Hz, NCH2CH2NHCO), 2.5 (4H, m, morpholine CH2N); δN (CDCl3): 69.16. δC (CDCl3): 165.25, 159.02, 140.82, 130.42, 130.07, 125.63, 125.57, 119.98, 119.76, 66.58, 57.01, 53.29, 35.90, 35.07. CIMS (NH3) m/z 319 (MH+−15NN, 3.3%), 291 (MH+−CH3NCO, 100). Anal. calcd for C16H22N6O3: C, 55.5; H, 6.4; N, 24.3. Found: C, 55.27; H, 6.33; N, 24.5.
13.
P. Skehan, R. Storeng, D. Scudiero, A. Monks, J. McMahon, D. Vistica, J.T.
Warren, H. Bokesch, H.S. Kenney and M.R. Boyd. J. Natl. Cancer Inst.
82 (1991), p. 1107.
| Bioorganic
& Medicinal Chemistry Letters Volume 10, Issue 20, 16 October 2000, Pages 2325-2327 |
|
|
 |